Biopharma Innovation, Testing Evolution, and Regional Growth Insights
The biopharmaceutical industry is entering a phase where innovation and commercialization are moving at unprecedented speed. The conversation is no longer limited to scientific breakthroughs. Decision makers now focus on scalability, regulatory strategy, and long-term investment outcomes. For companies navigating evolving therapeutic demand and rising R&D costs, structured market intelligence has become essential to identify where real growth is happening and where risks are emerging.
Below is a strategic overview of the current market landscape, highlighting key trends, opportunities, and areas where data-driven insights can support better planning.
A. Market Evolution: From Traditional Biologics to Advanced Therapies
The industry has shifted from conventional biologics toward complex, high-value therapies such as monoclonal antibodies, gene therapies, and advanced vaccines. Companies such Roche and Novartis continue to strengthen their monoclonal antibody portfolios, particularly in oncology and autoimmune treatments, contributing significantly to biologics revenue growth. Moreover, Pfizer and BioNTech have demonstrated how advanced vaccine platforms can accelerate innovation cycles, while Gilead Sciences and Moderna are investing heavily in gene and mRNA-based therapies that aim to transform long-term treatment models.
Innovation pipelines are expanding beyond therapeutic performance to include scalable manufacturing and personalized medicine strategies. Several leading firms are adopting modular production systems and data-driven clinical development to reduce timelines and improve commercialization outcomes. As competition intensifies and regulatory pathways evolve, companies increasingly rely on detailed competitive analysis to understand positioning, partnership opportunities, and market entry timing across different therapy segments.
B. Product Type Dynamics: Where Innovation Drives Revenue
Different product categories are progressing at varying speeds. Monoclonal antibodies and vaccines remain core revenue generators due to strong clinical validation and expanding application areas. Recombinant growth factors and purified proteins maintain steady demand, especially in metabolic and inflammatory disease management. Gene therapies represent a high-growth segment, driven by increasing investment and clinical breakthroughs. However, their commercialization challenges, including cost structures and regulatory scrutiny, make detailed market segmentation and forecast analysis critical for stakeholders evaluating entry strategies. This shift is also pushing companies toward platform-based development models rather than relying on single-product pipelines, enabling faster scalability and broader therapeutic expansion.
C. Service Landscape: Quality Testing and Compliance as Growth Enablers
Laboratory testing services are becoming central to biopharmaceutical development and commercialization. Customer proprietary testing and compendial or multi-compendial laboratory testing are expanding as companies prioritize data integrity and regulatory compliance. More than 60% of biopharmaceutical manufacturers depend on external contract testing organizations to manage complex analytical requirements and accelerate product release timelines. In addition, regulatory agencies continue to tighten quality standards, leading to a noticeable rise in demand for validated analytical methods and stability testing programs. Testing strategies are increasingly viewed not only as compliance requirements but also as critical risk-mitigation tools that support regulatory readiness, product safety, and faster global approvals.
D. Application Trends: Oncology and Autoimmune Segments Leading Demand
Oncology remains a primary revenue-driving segment within the biopharmaceutical landscape, supported by continuous advancements in targeted therapies and precision medicine approaches. Autoimmune, inflammatory, and infectious disease treatments continue to demonstrate steady expansion, driven by sustained healthcare investments and ongoing vaccine innovation. Moreover, neurological therapies are gaining strategic attention as research increasingly focuses on long-term disease modification rather than short-term symptom relief. Applications in metabolic and cardiovascular disorders maintain consistent demand, contributing to a balanced and diversified therapeutic portfolio. Evaluating adoption patterns across these applications enables organizations to refine pipeline priorities, optimize development strategies, and focus on high-value clinical opportunities.
E. Regional Outlook: Investment and Expansion Hotspots
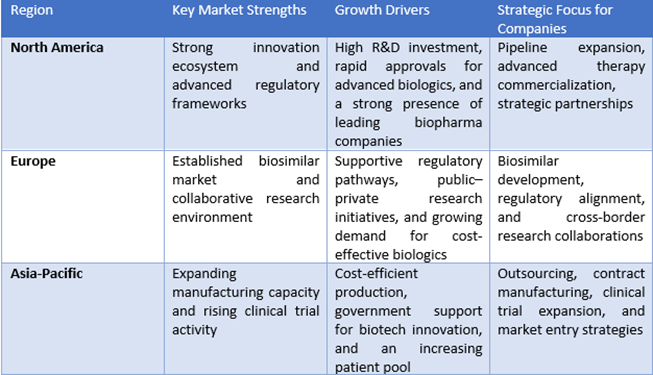
Source: OMRAnalysis
Union Budget 2025–26: Biopharma SHAKTI Initiative andPolicy Support
In February2026, the Union Budget 2025–26 introduced the Biopharma SHAKTI initiative, anational program with an allocation of ?10,000 crore over five years aimed atstrengthening India’s end-to-end biologics and biosimilars ecosystem. Theinitiative focuses on advancing domestic development and manufacturing ofhigh-value biopharmaceutical products, reducing reliance on imports, andimproving India’s position within global biologics supply chains. As part ofthe broader strategy, the government announced the establishment of three newNational Institutes of Pharmaceutical Education and Research (NIPERs) alongsidethe modernization of seven existing institutes to address the growing demandfor specialized talent across research, manufacturing, and regulatoryfunctions.
The budget alsoproposes the development of more than 1,000 accredited clinical trial sites toexpand India’s clinical research capabilities and accelerate innovation inadvanced therapies. In addition, regulatory strengthening measures, includingcapacity expansion at the Central Drugs Standard Control Organisation (CDSCO),are expected to improve approval efficiency and align evaluation timelines withglobal standards for complex biopharmaceutical products.